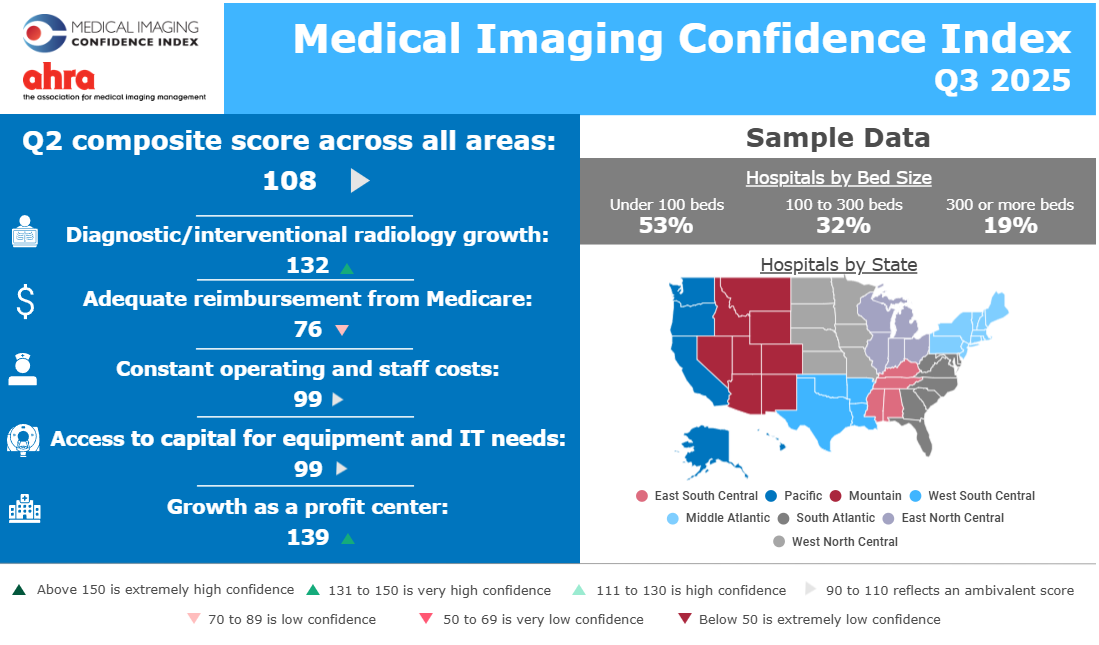
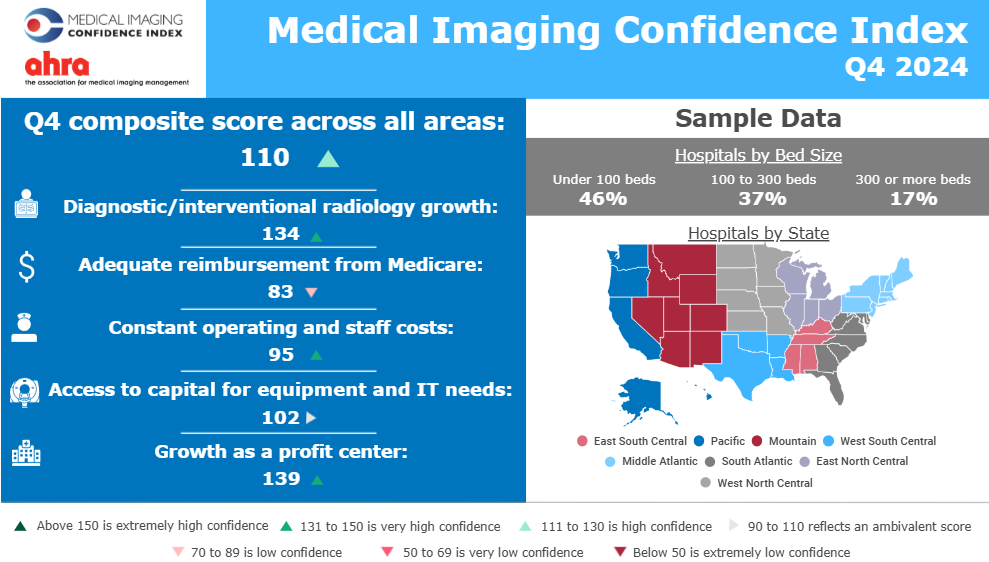
Media
Healthcare Insights from Professionals
We constantly leverage our extensive industry knowledge and expert relationships to reveal the latest trends across MedTech markets in order to bring you actionable news and insights.
Latest Articles


Contact Us
We would love to hear from you. Send a message via our contact page and we will get back to you as soon as we can.